Minimal information for studies of extracellular vesicles (MISEV2023): from basic to advanced approaches
About the MISEV Guidelines
The International Society for Extracellular Vesicles (ISEV) first published its “Minimal Information for Studies of Extracellular Vesicles,” in 2014 (MISEV2014) with an updated version published in 2018 (MISEV2018). ISEV are now proud to share the most recent version of the MISEV guidelines, MISEV2023.
The goal of MISEV 2023 is to provide researchers with an updated snapshot of available approaches and their advantages and limitations for production, separation, and characterization of EVs from multiple sources, including cell culture, body fluids, and solid tissues. In addition to presenting the latest state of the art in basic principles of EV research, MISEV 2023 also covers advanced techniques and approaches that are currently expanding the boundaries of the field. MISEV2023 includes new sections on EV release and uptake and a brief discussion of in vivo approaches to study EVs.
Compiling feedback from ISEV expert task forces and more than 1000 researchers worldwide, MISEV 2023 conveys the current state of EV research to facilitate robust scientific discoveries and move the field forward even more rapidly.
What was the process behind MISEV2023?
The achievement of publishing MISEV2023 is the culmination of a three-year process, summarised below. You can read more about this process in the concluding section of MISEV2023.
- 2020 pre-MISEV survey (summary article)
- ISEV board assigned a five-author committee (Welsh, Goberdhan, O’Driscoll, Théry, Witwer)
- Drafts and refinements with ISEV board input and invited drafting authors
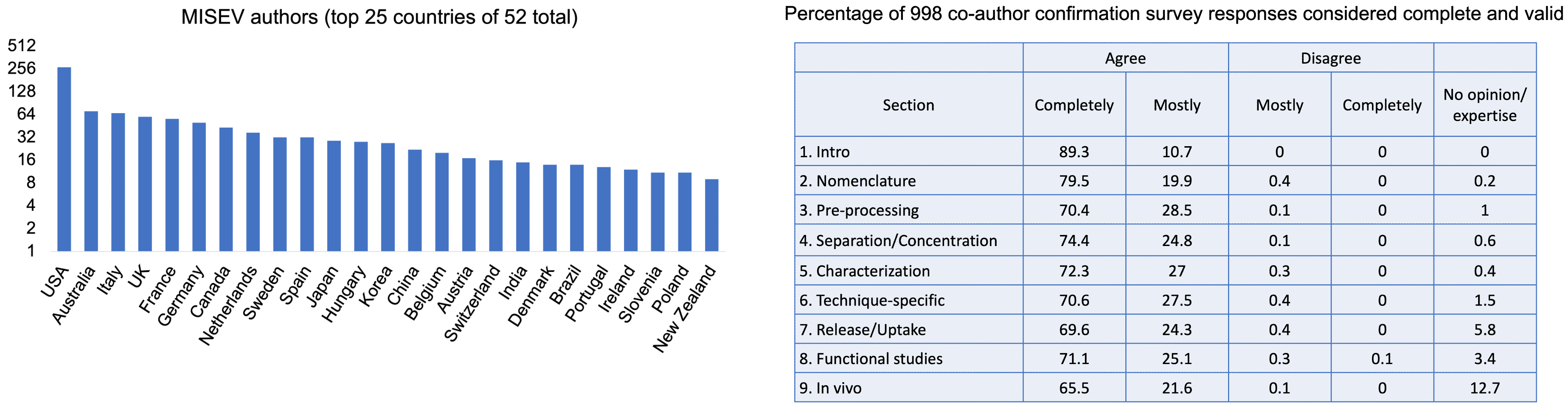
- 2022 co-author survey with >1000 responses
- Revisions based on the survey
- Internal review and additional invited contributions/revisions; style “unification”
- Journal pre-submission review and three post-submission reviews
- Co-author confirmation and consensus survey
MISEV2023: domains
- Nomenclature: communication of concepts
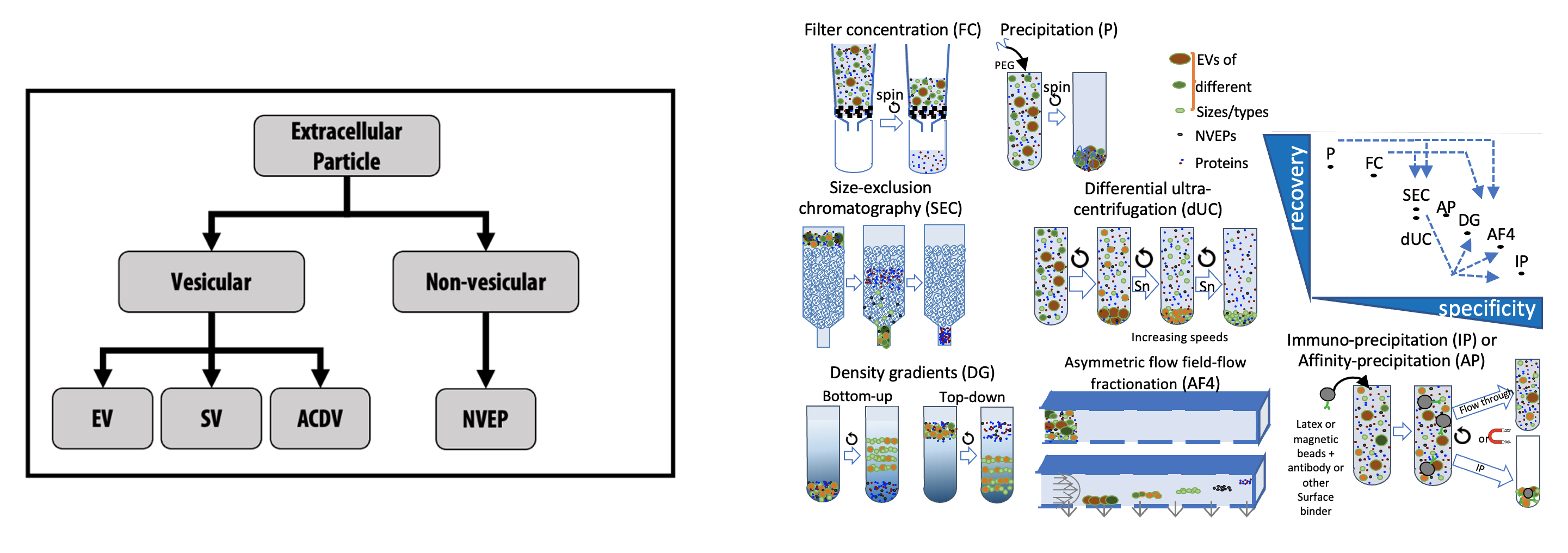
- How to approach Non-Vesicular Extracellular Particles
- Collection and pre-processing: pre-analytical variables: EV sources including bacteria, biofluids, tissue
- EV separation and concentration
- EV characterization
- Technique-specific reporting for EV characterization
- EV release and uptake
- Functional studies
- In vivo EV studies
New sections in MISEV2023 are highlighted in bold.
MISEV2023: 1051 co-authors in 53 countries
What MISEV is NOT…or should not be…
- An unreasonable barrier to field entry
- Rather, a guide to doing rigorous and publishable science
- An attempt to stifle innovation
- See the MISEV2018 section on “exceptions”; MISEV2023 “IS NOT” section
- Irrelevant to non-mammalian EV studies
- Principles of good definitions, markers, and controls are relevant to all EV sources. The exact markers and procedures will vary.
- Irrelevant to clinical studies: biomarkers, therapeutics
Extracellular particle (EP) nomenclature and separation/concentration methods
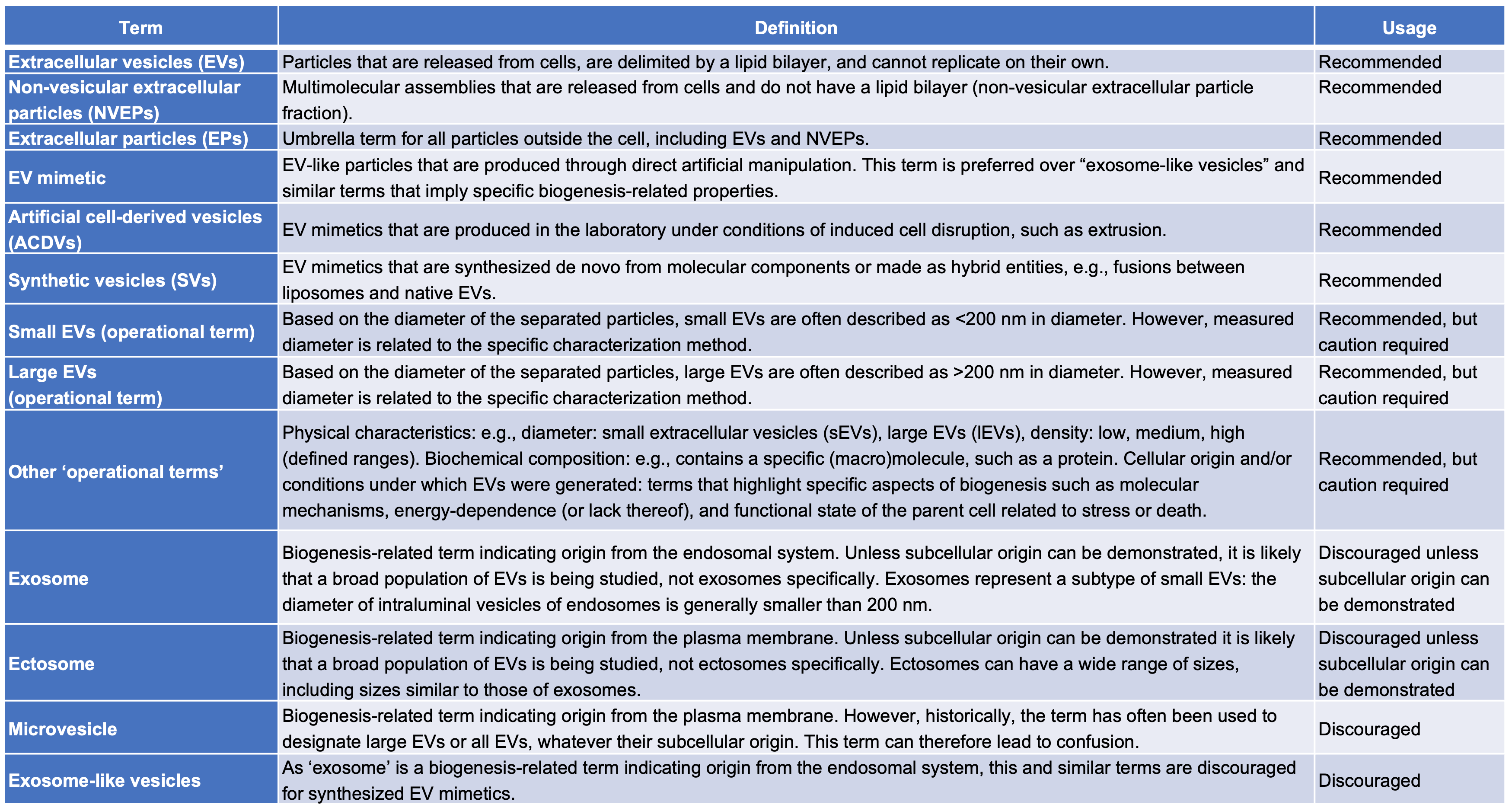
Quick reference on EV terms
Spread the Word!
Thanks to community members for featuring MISEV2023. Read more:
- Nadeau and Burnier, "With >1000 authors, the updated “Minimum Information for Studies of Extracellular Vesicles” (MISEV) promotes rigor, reproducibility, and transparency in the liquid biopsy field" The Journal of Liquid Biopsy, 2024
- Samuels and Giamas, "MISEV2023: Shaping the Future of EV Research by Enhancing Rigour, Reproducibility and Transparency", Cancer Gene Therapy, 2024
- Poupardin, et al, "Advances in Extracellular Vesicle Research Over The Past Decade: Source And Isolation Method Are Connected with Cargo And Function", Advanced Healthcare Materials, 2024
Please engage and help us spread the word about this valuable resource.

If you are able to help, please complete the questionnaire. Thank you for your support of MISEV and ISEV!
|

This document has been put together based on the questions we have been asked since we announced the acceptance of MISEV2023 for publication.
|

The PowerPoint describes the process behind MISEV2023 as well as some of the other ISEV initiatives.
|

Keep an eye on the evolution of recommendations by ISEV Rigor and Standardization Subcommittee, such as Task Forces, Position papers and guidelines, etc. Do you have a suggestion? Want to join? Contact us!
|

- Subscribe today!
- >4300 subscribers
- Educational videos, Plenary talks, ISEV2023, MOOC3, etc.
|
 |
|